
cell processing
Certified
Stem Cell &
Genetic Laboratory
Sourcing & Processing Autologous Stem Cells from Adipose Tissue
Expansion & Culture of Autologous Stem Cells
Differentiating Adipose Stem Cells into Induced Pluripotent Stem Cells (iPSCs)
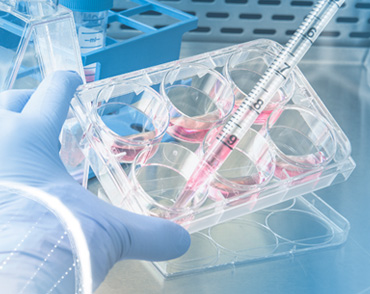
Stem Cell Laboratory Processing
The proper medical term for the process of collecting, preparing, formulating, producing, and expanding stem cells is “stem cell processing.” This term encompasses the various steps involved in obtaining and manipulating stem cells for therapeutic purposes. Stem cell processing typically involves techniques such as cell isolation, purification, culture, expansion, and differentiation, depending on the specific goals of the procedure.
Procedure for Sourcing Autologous Stem Cells from Adipose Tissue:
Adipose Tissue Collection: The procedure begins with the collection of adipose tissue from the patient’s body. This is typically done through a minimally invasive technique called liposuction. A small incision is made, and adipose tissue is gently suctioned out using a specialized cannula.
Tissue Processing: The collected adipose tissue undergoes processing to isolate the stem cells within. Various enzymatic or mechanical methods can be employed to break down the tissue and release the stem cells. This step aims to separate the stem cells from other components, such as fat cells and connective tissue.
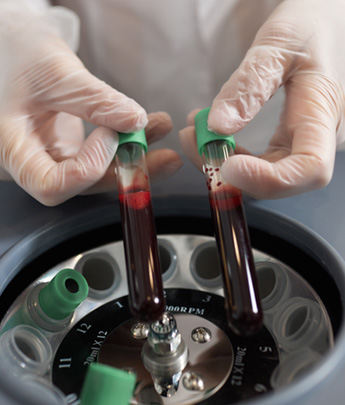
Stem Cell Isolation: Once the adipose tissue is processed, the next step is to isolate the stem cells from the mixture. Techniques like centrifugation or filtration are commonly used to separate the stem cells based on their density or size.
Stem Cell Concentration: After isolation, the stem cells are concentrated to obtain a higher number of viable cells. This can be achieved through additional centrifugation or filtration steps, removing excess fluids and debris while retaining the stem cells.
Quality Control: Prior to further processing, a quality control assessment is conducted to evaluate the viability, purity, and potency of the isolated stem cells. This may involve testing for specific markers, cell count, viability assays, and sterility checks.
Procedure for Expansion and Culture of Autologous Stem Cells:
Cell Culture Preparation: The isolated autologous stem cells are placed in a sterile culture vessel containing a suitable growth medium. The growth medium is formulated to provide the necessary nutrients, growth factors, and support for cell proliferation and maintenance.
Cell Expansion: The stem cells are allowed to proliferate and expand in the culture vessel under controlled laboratory conditions. This involves providing an optimal environment with appropriate temperature, humidity, and gas composition.
Medium Changes: At specific intervals, the growth medium is replaced or supplemented to support cell growth and maintain an optimal cell culture environment. This ensures the provision of fresh nutrients and removal of waste products.
Passage and Subculturing: As the stem cells multiply, they may reach confluency in the culture vessel. At this stage, they need to be passaged or subcultured to provide sufficient space for continued growth. This involves detaching the cells from the vessel surface, typically using enzymatic or mechanical methods, and transferring them to new culture vessels.
Expansion Monitoring and Control: Throughout the expansion process, the stem cells are regularly monitored for growth kinetics, morphology, viability, and other quality parameters. This allows for adjustments in culture conditions, medium composition, and passaging techniques, as needed, to maintain optimal cell health and growth.
Procedure for Differentiating Adipose Stem Cells into Induced Pluripotent Stem Cells (iPSCs):
Reprogramming:
a. ASCs are transduced with viral vectors or transfected with plasmids carrying reprogramming factors.
b. The commonly used reprogramming factors include OCT4, SOX2, KLF4, and c-MYC.
c. These factors can be introduced using viral vectors (such as lentivirus or retrovirus) or non-integrating methods like mRNA or proteins.
d. The reprogramming factors act to reset the cellular state and induce pluripotency in the ASCs.Colony Formation and iPSC Selection:
a. The reprogrammed cells are plated at low densities to form colonies.
b. Individual colonies are manually picked or mechanically isolated using micropipettes or automated systems.
c. The selected colonies are transferred to fresh culture plates or dishes for further expansion and characterization.iPSC Culture and Expansion:
a. The selected iPSC colonies are maintained and expanded in an appropriate culture medium that supports pluripotency.
b. The medium contains specific growth factors, such as basic fibroblast growth factor (bFGF) or leukemia inhibitory factor (LIF), which help sustain the pluripotent state.
c. Regular passaging and subculturing are performed to maintain the iPSC population and prevent spontaneous differentiation.iPSC Characterization:
a. The iPSCs are characterized to confirm their pluripotent nature and genetic stability.
b. Techniques such as immunostaining, flow cytometry, and gene expression analysis are used to assess the expression of pluripotency markers (e.g., OCT4, NANOG, SSEA-4, TRA-1-60, TRA-1-81) and the absence of lineage-specific markers.
c. Karyotyping or genetic analysis is performed to ensure the stability of the iPSC genome.The selected iPSC colonies are further characterized to confirm their pluripotent nature. This includes analyzing the expression of pluripotency-associated genes (e.g., OCT4, NANOG, SOX2) using techniques like immunocytochemistry or quantitative polymerase chain reaction (qPCR). Additionally, the iPSCs are evaluated for their ability to form embryoid bodies (3D aggregates representing different germ layers) and their potential to differentiate into various cell types representing the three germ layers, a process known as in vitro differentiation.
It is important to note that these treatments are still considered experimental and have not yet received approval from Western medical standards. While there have been promising results and anecdotal evidence of positive outcomes, there is currently no definitive scientific proof of a cure for any specific condition using stem cell therapy. It is crucial to understand that what works for one patient or in a small sample size may not necessarily be applicable to others or prove effective in larger clinical trials. Patients considering stem cell therapy should be aware of the experimental nature of these treatments and exercise caution when evaluating potential benefits.